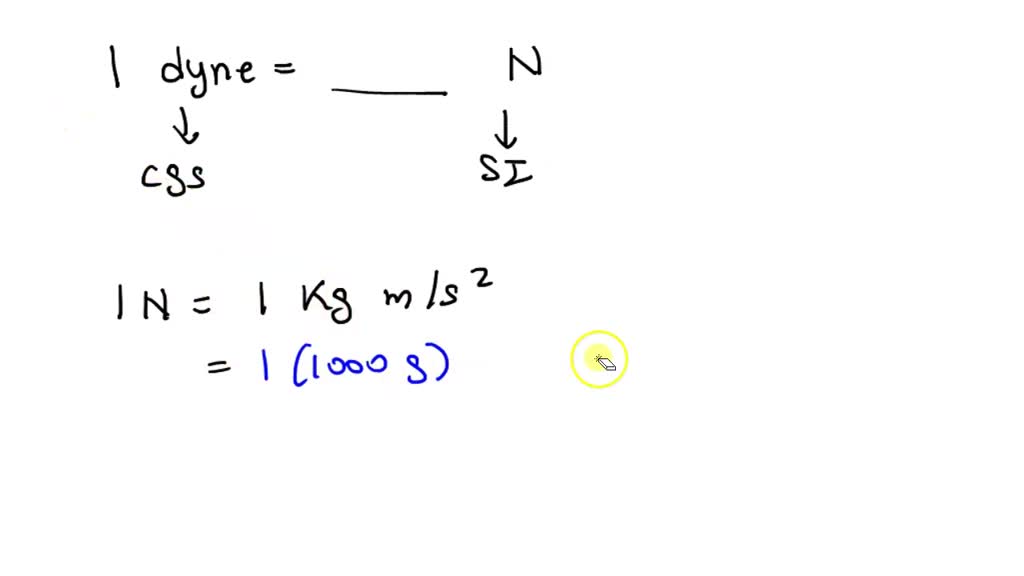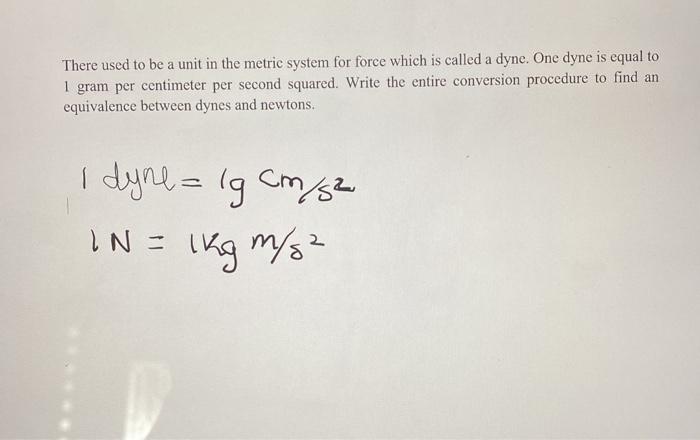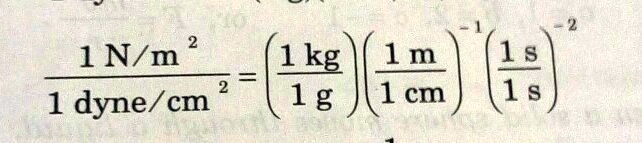
SOLVED: 'solve it plz conversion factor between newton and dyne 1 N/m 1 dyne/ cm 1kg 1n | (4 1 g cm 1 '
The value of G in C.G.S system is 6.67 × 10^-8 dyne cm^2 g^-2. Calculate its value in S.l. system. - Sarthaks eConnect | Largest Online Education Community

Refugia map for G. pulex, indicating zones below 30 dynes (dyne/cm 2 )... | Download Scientific Diagram
![Canadian Preparation H Cream with Bio-dyne Large 50 Gram size. Bio-Dyne, a natural yeast cell extract, is The Models' secret for younger looking eyes. by Pfizer [Beauty] : Amazon.ca: Health & Personal Canadian Preparation H Cream with Bio-dyne Large 50 Gram size. Bio-Dyne, a natural yeast cell extract, is The Models' secret for younger looking eyes. by Pfizer [Beauty] : Amazon.ca: Health & Personal](https://images-na.ssl-images-amazon.com/images/I/416K9c7aGmL._AC_UL200_SR200,200_.jpg)
Canadian Preparation H Cream with Bio-dyne Large 50 Gram size. Bio-Dyne, a natural yeast cell extract, is The Models' secret for younger looking eyes. by Pfizer [Beauty] : Amazon.ca: Health & Personal
The value of G in C.G.S system is 6.67 × 10^-8 dyne cm^2 g^-2. Calculate its value in S.l. system. - Sarthaks eConnect | Largest Online Education Community

If at a pressure of 10^6 dyne/cm^2 , one gram mole of nitrogen occupies 2 × 10^4 c.c. volume, then the average energy of nitrogen molecules in erg is :

The value fo universal gravitationla constant `G` in `CGS` system is `6.67xx10^(-8)` dyne `cm^(2) - YouTube

Activity 1: solve for size and speed of a drop.. Useful units for calculations Remember: dyne = 1 g cm/s 2 (force in cm-g-s units) = Newtons. Using. - ppt download